Higher and foundation tiers

The rate of a chemical reaction tells us how fast it is going. Some reactions such as a firework exploding are very fast while other reactions such as the fermentation of sugar to make wine or the rotting of pears may take weeks while the rusting of iron is a chemical reaction that may take years. Other chemical reactions such as the digestion of food may only take a matter of hours to complete.
There are many ways to measure the rate of a reaction; you could for example measure:

We could measure the rate of this reaction by measuring how quickly one of the reactants is used up or how quickly one of the products is produced.
That is how quickly the solid
calcium carbonate or the hydrochloric acid is used up. However one of the products is the gas carbon dioxide. It would
be much
easier to measure the rate of reaction
by measuring how quickly this gas is given off. There are a number of ways of doing this.
One way to measure the rate of this reaction is to time how quickly the gas carbon dioxide is released. The apparatus shown below could be used to do this. You could use the gas syringe to measure how much carbon dioxide gas is given off every 30 seconds until the reaction stops.

Using the method shown above you could measure the rate of reaction by measuring the volume of carbon dioxide gas released every 30 seconds. Obviously the larger the volume of gas released the faster the reaction. The rate of reaction could be calculated by simply dividing the volume of gas released by the time taken. A typical set of results for this experiment is shown in the table below:
| time/s | 0 | 30 | 60 | 90 | 120 | 150 | 180 | 210 | 240 |
|---|---|---|---|---|---|---|---|---|---|
| volume of gas/cm3 | 0 | 40 | 75 | 100 | 111 | 116 | 119 | 119 | 119 |
 A graph of these results was drawn and is shown opposite. There are a few points you should note from the graph:
A graph of these results was drawn and is shown opposite. There are a few points you should note from the graph:
This method is very similar to the one above. You probably used one or other method in your science lessons. Below an inverted measuring cylinder is filled up with water. As the reaction starts the carbon dioxide gas passes down the delivery tube and rises up into the measuring cylinder pushing the water out as it does. You can measure the volume of gas in the measuring cylinder every 30 seconds and calculate the rate of reaction from your results. This method works well for carbon dioxide gas since its solubility in water is fairly low. Obviously this method would not suitable if the gas to be collected was very soluble in water.

Since carbon dioxide gas is a heavy gas it is possible to measure the rate of this reaction by measuring the loss in mass
as the reaction takes place. In the apparatus below the carbon dioxide gas released by the reaction
will escape through the pipe in the stopper. It is essential to have some kind of stopper as the effervescence (fizzing) from the
bubbling can cause splashes which if they left the conical flask would affect the results.
A reading of the mass on the balance could be
taken every 30 seconds and this change in mass could be used to calculate the rate of the reaction. A valid set of results should show that to begin with there will be a large drop in mass as large
amounts of carbon dioxide is released. As the reaction proceeds there will be less reactants available to react so less carbon dioxide gas should
be released; this means the mass will drop more slowly. This method would not be particularly suitable for lightweight gas
such as hydrogen.

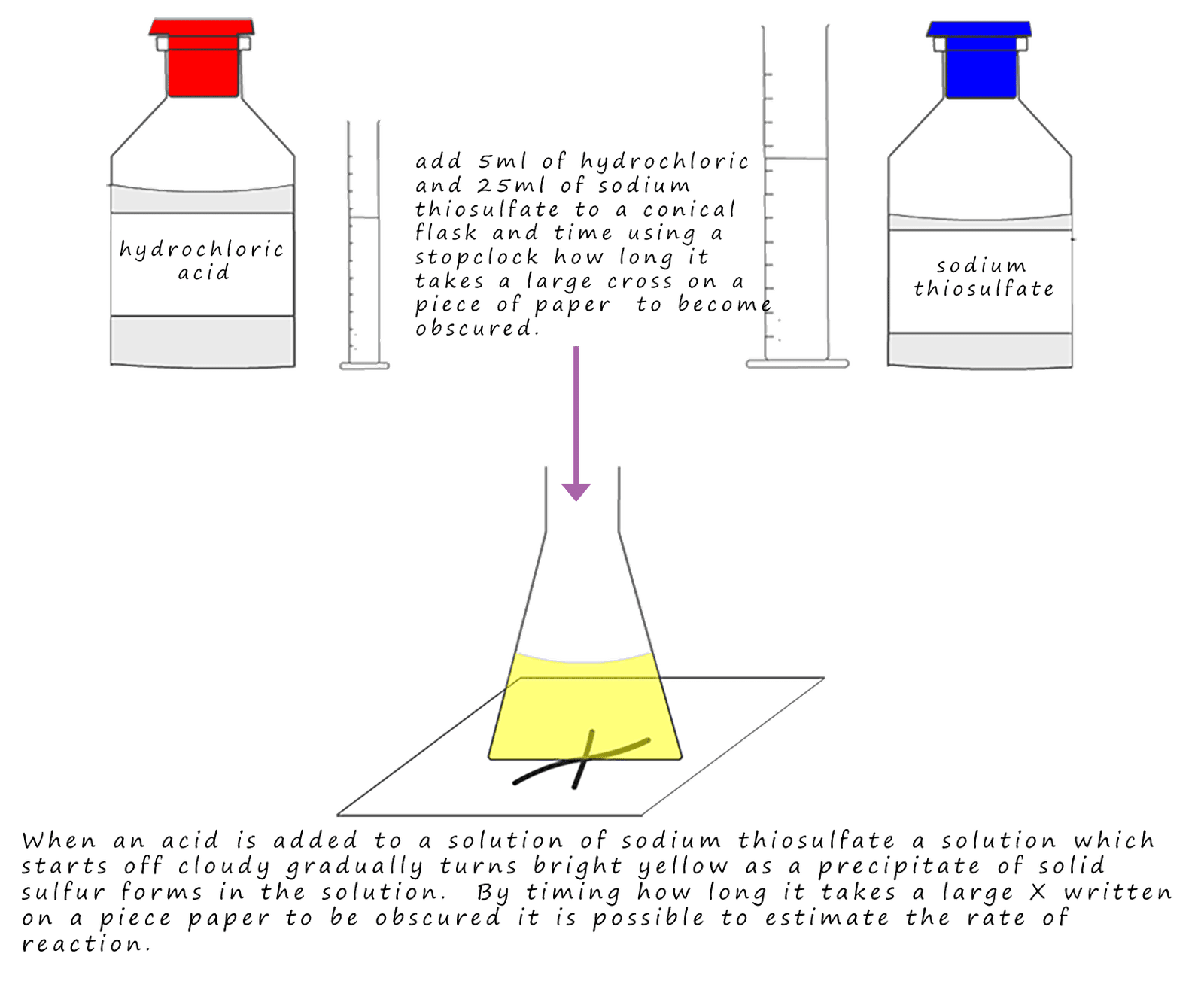
Consider the reaction between sodium thiosulfate and hydrochloric acid:

One of the products of this reaction is the gas sulfur dioxide; so we could measure the rate of reaction as before by
measuring how much gas is given off in certain time period using a gas syringe. However sulfur dioxide is a toxic gas and so
it is probably not advisable to collect large amounts of it in a syringe. All of the reactants: sodium
thiosulfate and hydrochloric acid are colourless solutions; similarly water and sodium chloride solution on the products
side of the above equation are also colourless. This leaves sulfur; sulfur is a yellow solid that is insoluble in water.
In this
reaction as the solid sulfur is produced it causes the solution to gradually turn yellow. That is it changes colour.
This means it is possible to measure the rate of this reaction by timing how long it takes the solution to change colour or turbidity. This is usually done by timing how long it takes a large cross (X) drawn on a piece of paper to become obscured by the solid sulfur produced in the reaction. The procedure for this is outlined in the image below: