Chemistry only
Testing for cations
Cation ions are positively charged ions, mostly metals ions. They are
ions that would be attracted to a
negatively charged cathode, hence cation.
Flame tests
Most people remember doing this practical; probably because you get to burn stuff! Some metal ions
will give a very distinctive colour when placed in a hot flame. A loop of wire; usually platinum is
placed in some concentrated acid to make sure it is clean and free from any residues that could
contaminate the flame and give a false result. Once it is clean hold it in a
hot flame for a few
seconds until only the blue colour of the flame is visible. Next dip the platinum wire into a solution
containing the metal ion you hope to identify, then insert the wire loop back into the hot Bunsen flame
and record any colour you observe. The flame colours for some
metal ions are shown below:
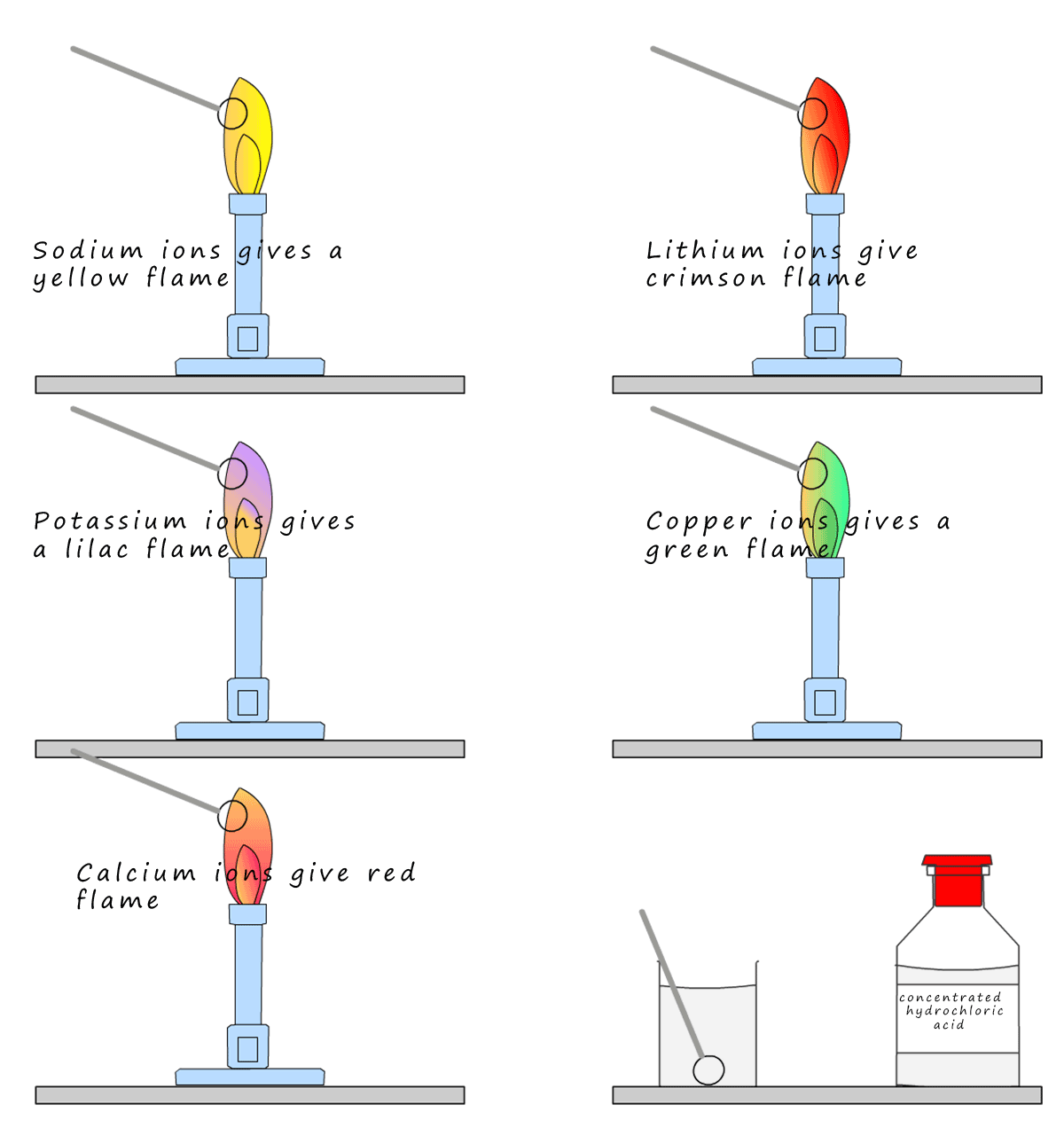
The only real problem with flame tests is that if more than one
metal ion is present then you can
get mixed results where it is hard to tell exactly what metal ion maybe present and also the fact that flame test gives no indication of the amount of the metal ion present. The main reason for this
is some of the colours are more intense and
over powering than others. The yellow sodium flame is very intense and will mask other colours, as is the red lithium flame
colour.
Precipitation reactions
Some transition metal ions form a coloured precipitate when an alkaline solution such as
sodium hydroxide is added. The colour of the precipitate formed can help you identify unknown metal ions.
A precipitate is an insoluble solid formed when 2 solutions are mixed e.g.
A(aq) + B(aq) → C(s) + D(aq)
Here you need to look at the state symbols; (aq) is for a soluble solution and (s) is for an insoluble solid.
Here two solutions A and B are mixed and an insoluble solid C is formed. This
insoluble solid could be
removed by filtering.
Most solutions of metal ions with a 2+ and 3+ charge will form insoluble precipitate when an
alkaline
solution such as sodium or potassium hydroxide is added to them.
The image below shows 4 boiling tubes each containing a solution of a metal ion with a 2+ or
3+ charge. A few drops of sodium hydroxide was then added to each of the boiling tubes and as you can see from
the image below a coloured precipitate is formed in each boiling tube. Equations for each of these reactions is given below:

Most metal ions with a 2+ and 3+ charge will form insoluble precipitate when an
alkaline
solution such as sodium or potassium hydroxide is added.
Example 1: Precipitates from copper(II) ions (Cu2+)
Copper(ll) sulfate(aq) + sodium hydroxide(aq) → sodium sulfate(aq) + copper hydroxide(s)
CuSO4(aq) + 2NaOH(aq) → Na2SO4(aq) + Cu(OH)2(s)
An ionic equation for this reaction is:
Cu2+(aq) + 2OH-(aq) → Cu2+ (OH-)2(s)
All sodium compounds are soluble so the insoluble solid produced must be copper hydroxide. Copper hydroxide is the
blue solid precipitate produced.
Example 2: Precipitates from iron (II) ions (Fe2+)
Word, symbolic and ionic equations for the formation of iron(ll) hydroxide:
Iron (ll) chloride(aq) + sodium hydroxide(aq) → sodium chloride(aq) + iron(ll) hydroxide(s)
FeCl2(aq) + 2NaOH(aq) → 2NaCl(aq) + Fe(OH)2(s)
The solid precipitate of iron hydroxide produced is a green colour. An ionic equation for this reaction can be written, it would also make sense to remove all the spectator ions,
that is ions which are unchanged on both the reactant and product sides of the equation. These ions are only there to balance off the charges.
Fe2+(aq) + 2OH-(aq) → Fe2+(OH-)2(s)
Example 3: Word, symbolic and ionic equations for the formation of solid iron(lll) hydroxide:
Iron(lll) chloride(aq) + sodium hydroxide(aq) → sodium chloride(aq) + iron(lll) hydroxide(s)
FeCl3(aq) + 3NaOH(aq) → 3NaCl(aq + Fe(OH)3(s)
Fe3+(aq) + 3OH-(aq) → Fe3+(OH-)3(s)
The solid precipitate of iron (lll) hydroxide produced is a brown colour.
Testing for Al3+, Ca2+ and Mg2+ ions
All the examples above use transition metals but with a little more chemistry it is possible to use
this reaction to help identify calcium ions (Ca2+), magnesium ions (Mg2+), and
aluminium ions (Al3+).
If a sodium hydroxide solution is added to a
solution of these ions then
a white (colourless) precipitate of the metal hydroxide is formed in each case.
Ca2+(aq) + 2OH-(aq) → Ca2+(OH-)2(s)
Mg2+(aq) + 2OH-(aq) → Mg2+(OH-)2(s)
and for aluminium:
Al3+(aq) + 3OH-(aq) → Al3+(OH-)3(s)
This might not seem particularly helpful in identifying these three ions if they all produce 3 colourless (white) solid precipitates.
However:
- If an excess of sodium hydroxide is added to
aluminium hydroxide in a test-tube the white precipitate
now becomes soluble again and the solution becomes clear, this does not happen with calcium or
magnesium hydroxides.
This simple test will enable you to test for the presence of Al3+ ions. Now all we need is
a simple way
to distinguish calcium ions from magnesium ions. This is easily done:
- Calcium ions can be distinguished from magnesium ions by
a simple flame test. Calcium ions produce an orange -red flame colour during a flame test whereas
magnesium ions produce no colour during a flame test.
Test for ammonium ions
Ammonium ions (NH4+) can be detected in a substance by testing for the presence of ammonia gas (NH3)
which is released when ammonium ions are heated gently with a strong alkali such as sodium or potassium hydroxide.
NH4+(aq) + OH- (aq) → NH3(g) + H2O(l)
The ammonia gas which is released can be detected by using a strip of damp red litmus paper which will turn blue in
the presence of the basic ammonia gas. The test is outlined below:

Key points
- Flame tests can be used to identify many metal ions. The metals give different colours when placed in a
hot
Bunsen flame.
- Transition metal ions often form coloured precipitate when in alkaline solutions. The colour of these precipitates can be used
as a quick way to identify the metal present e.g. Cu2+
ions produce a blue precipitate, Fe2+ ions
produce a green precipitate and Fe3+ ions produce a
red/brown precipitate.
- Ammonium ions can be detected due to the fact that ammonia gas is released when ammonium ions are warmed with a
strong alkali.
Practice questions
Next




