

Chemistry only
 One of the problems with cells and batteries is that the chemicals inside them that react to produce
useful electrical energy eventually run out and the cell/battery has to be disposed of;
that is unless the cell is of a type which can be recharged.
However even rechargeable cells
can only be recharged so many times before they need replacing.
One of the problems with cells and batteries is that the chemicals inside them that react to produce
useful electrical energy eventually run out and the cell/battery has to be disposed of;
that is unless the cell is of a type which can be recharged.
However even rechargeable cells
can only be recharged so many times before they need replacing.
A potential solution to this problem is to use a fuel cell. A fuel cell is similar to an ordinary cell/battery but the chemicals which react to produce electrical energy are not contained inside the cell or battery but are supplied continually from an external source. This means that unlike a battery or a cell; as long as the reacting chemicals are supplied continuously then the fuel cell will keep producing electricity.
There are several different types of fuel cells but they all have many similarities in common.
A fuel; commonly hydrogen, alcohol or methane is supplied continually to the fuel cell.
All fuel cells contain an anode,
a cathode and an electrolyte.
In cells and batteries the anode has a negative charge and the
cathode is positively charged; this is the
other way round from what you will have met before in electrolysis where the anode has a positive charge and the cathode has a negative charge, so be careful not to mix them up!
The anode catalyst is commonly platinum and the cathode catalyst is commonly made from the metal nickel. The anode, cathode and electrolyte are kept apart in a fuel cell by a semi-permeable membrane. This membrane allows ions to move across it but not electrons. The electrons are forced through an external circuit (a wire) where they are used to power whatever is attached to the fuel cell; this could be for example: a car, a motorcycle or some sort of electrical device or appliance. The diagram below shows an outline of the main parts of a typical hydrogen- oxygen fuel cell.
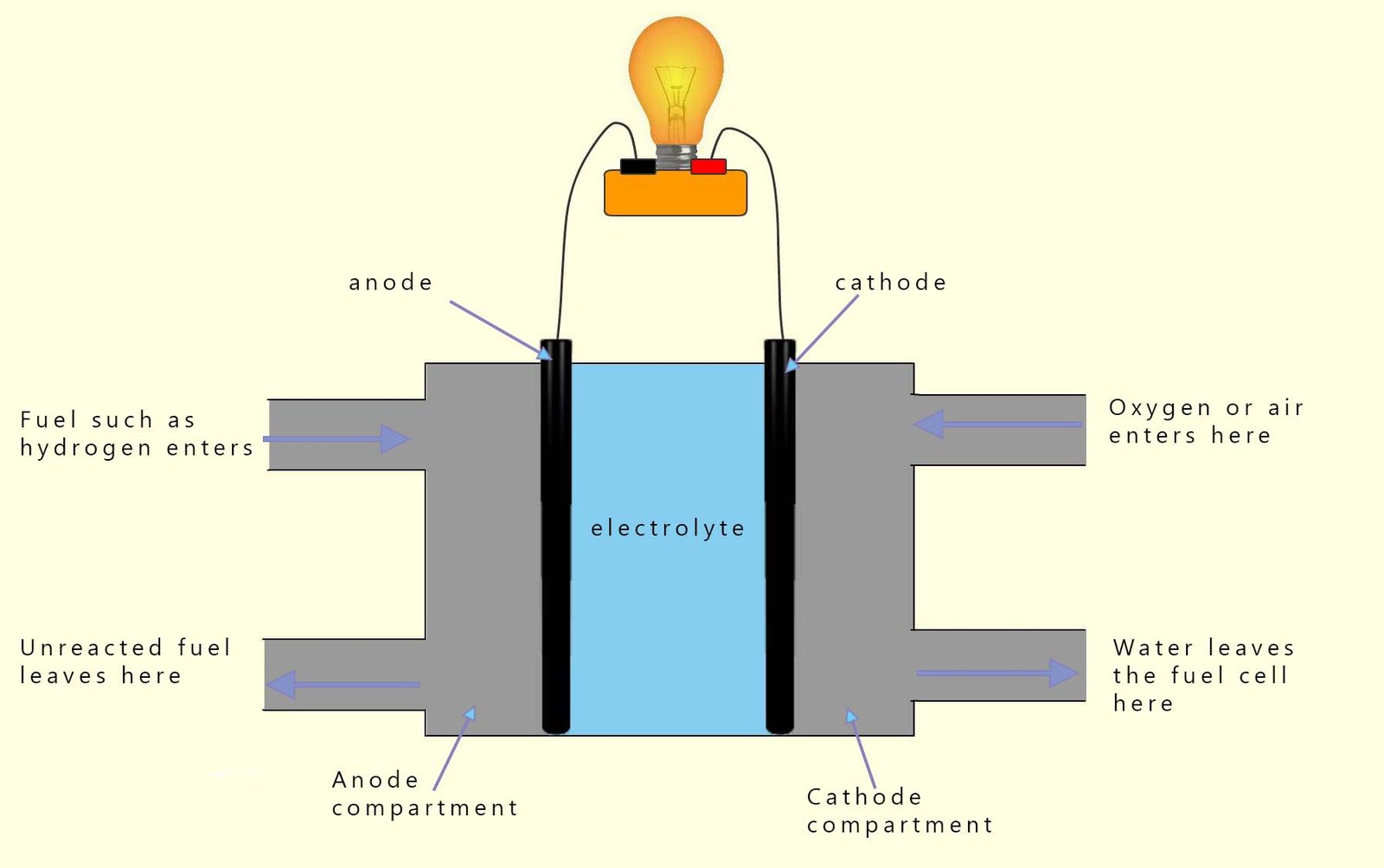
The diagram above shows an outline of a typical hydrogen-oxygen fuel cell. In this cell the hydrogen gas (the fuel) enters the porous carbon anode. Here the hydrogen is oxidised by the platinum catalyst present on the anode surface to form hydrogen ions (H+). This oxidation reaction can be shown as:
In a hydrogen fuel cell, oxidation and reduction happen at different electrodes — just like in electrolysis.
🧠 Exam memory tip:
Oxidation at the anode,
reduction at the cathode — every time.
OIL RIG →
Oxidation Is Loss (of electrons)
Reduction Is Gain (of electrons)
The electrons produced at the anode then flow through the wire connected to the bulb (or any electrical item). The hydrogen ions (H+) diffuse from the anode across the semi-permeable membrane and move through the electrolyte towards the cathode. At the cathode oxygen gas (O2) (from the air) is fed in and undergoes a catalysed reaction on the cathode surface to form water. The oxygen gas (O2) reacts with the hydrogen ions (H+) and the electrons arriving via the external circuit or wire. At the cathode the oxygen gas is reduced at the cathode to form water. An equation for this reduction reaction is:
The overall equation is obtained by combining the anode and cathode half-equations. We multiply the anode half-equation by two to balance the electrons and ensure there are 4e on both sides of the two half-equations.
The overall cell equation for this redox reaction is shown below, recall that: oxidation occurs at the anode and reduction at the cathode.
A fuel cell operates at various temperatures, usually between about 50 and 150oC, and many hydrogen-oxygen fuel cells can convert the stored energy in fuels into electricity with an efficiency often reaching about 40 to 60%.
The image below shows how a fuel cell works. Starting at the left hand-side, the anode side we have:
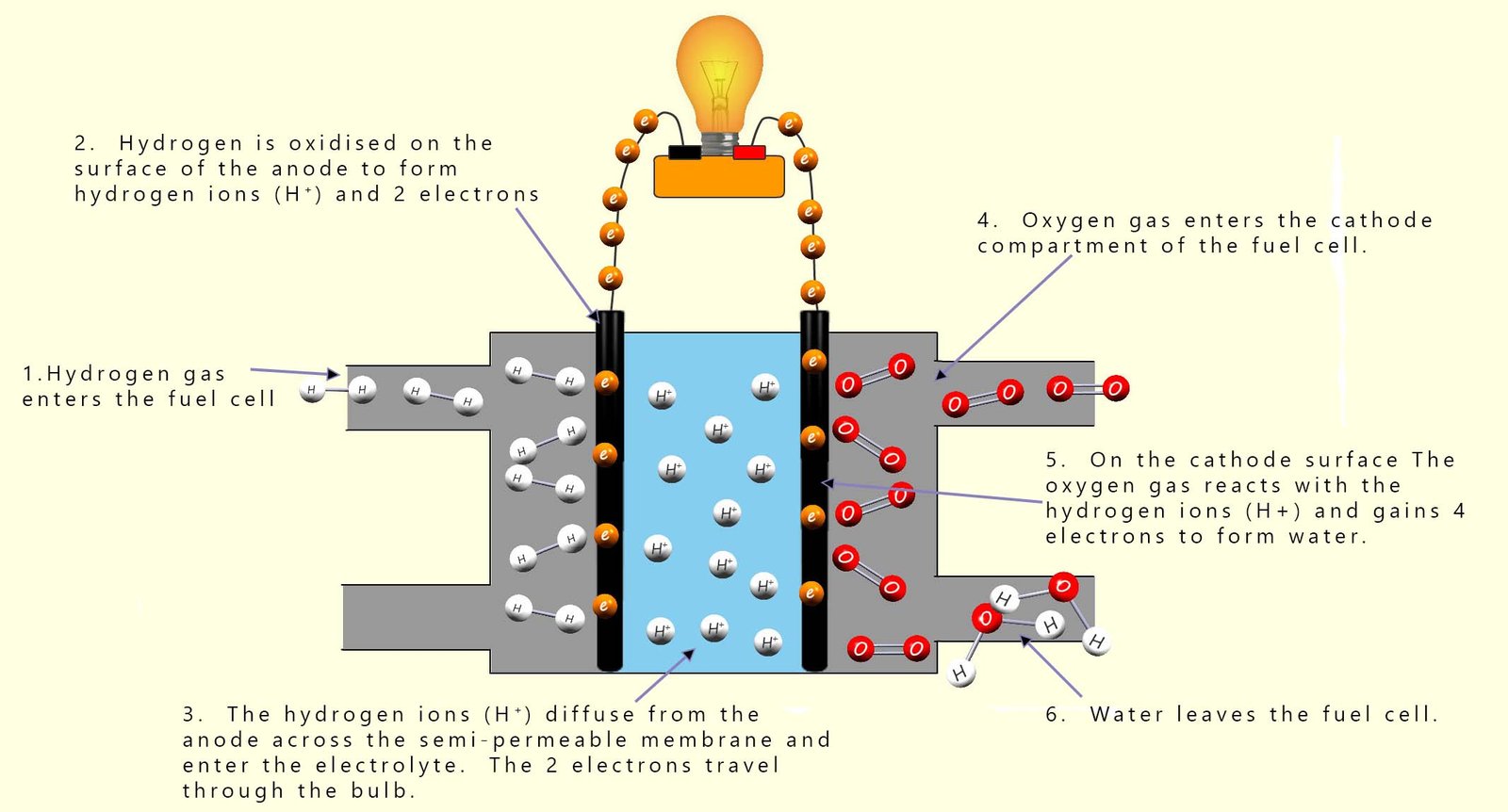
On the right-hand side of the fuel cell is the cathode where:
Try the quick activity below to review your understanding of the half-equations for the reactions taking place at the anode and cathode in a fuel cell.
Tap the pieces to build each equation. Tip: if you add the wrong piece, just tap it in the built line to remove it 🔁
Try the quick quiz below to review your understanding of how a hydrogen-oxygen fuel cell works.
Tap Anode or Cathode for each statement. Then hit Check to see how you did ✅
Fuel cells are incredibly flexible pieces of technology because they can be built in a wide range of sizes and designs that can produce a wide range of power outputs. At one end of the scale large fuel cell systems can generate electricity for factories, offices and even entire housing developments. These larger fuel cell systems are sometimes used as backup power supplies for hospitals or data centres where a reliable electricity supply is absolutely essential.
At the other end of the scale, fuel cells can be made small enough to power portable devices such as laptops, radios and phone chargers. Since fuel cells keep producing electricity for as long as they are supplied with fuel they can last much longer than a typical battery before needing to be refuelled.
One of the most well-known applications of fuel cells is in transport. Several car manufacturers; including Honda, Toyota and Hyundai have developed hydrogen fuel cell cars. One of the main advantages of these vehicles is that they generate their own electricity onboard and produce only water as a waste product.
Fuel cells are particularly useful where a combination of long range, fast refuelling and low emissions is important. This is why they are increasingly being explored for use in buses, trains, heavy goods vehicles and even boats.
Fuel cells are not just an experimental technology; they are already being used in many different sectors. The table below summarises some of the most important applications and explains why fuel cells are particularly suitable in each case.
| Application | How They’re Used |
|---|---|
| 🚍 Buses | Hydrogen-oxygen fuel cell buses are already running in some cities such as London. These buses can travel 300+ miles and refuel in about 10 minutes, which is a refuelling to a similar diesel powered bus. |
| 🚆 Trains | Used on regional and commuter lines as a cleaner alternative to diesel; for example hydrogen powered trains would be ideal for long-distance, rural routes such as the Scottish Highlands or parts of Wales where installing overhead electric wires is too expensive. |
| 🚛 Other Vehicles | Hydrogen-oxygen fuel cells are also found in heavy trucks, vans, taxis and forklift trucks. |
| 🚢 ✈️ Marine & Air | Hydrogen-oxygen fuel cell technology is also expanding into boats, ferries and drones. |
| Benefit | Why It Matters |
|---|---|
| 💧 Zero Emissions | The only waste product is water. No carbon dioxide is produced while running. |
| ⛽ Fast Refuelling | Refuelling takes just a few minutes and is much faster than charging a battery powered electric vehicle. |
| ❄️ Strong Performance | Work well in cold weather and is also suitable for heavy-duty use. |
Fuel cells can be built in many sizes and can produce very different power outputs.
One of the biggest challenges with fuel cells is the fuel itself ;the hydrogen gas. Hydrogen is an excellent fuel which releases lots of energy and produces only water💧 as a waste products; but it is not widely available in large amounts, so it has to be produced.
A common way of producing hydrogen is by splitting water using electrolysis, but this requires a lot of energy. If that energy comes from fossil fuels then the overall carbon footprint can still be high. Hydrogen can also be made from methane (CH4), but methane (CH4) is a fossil fuel so this method of obtaining hydrogen does not solve the long-term sustainability problem, that is fossil fuels are a non-renewable energy resource. In both these cases the energy used to create the hydrogen gas ideally needs to come from renewable sources.
However at the moment it is unlikely that there are enough renewable energy resources available to produce enough hydrogen to quickly replace all petrol and diesel vehicles.
Fuel cells are generally more expensive than batteries. This is mainly because they use costly catalysts at the anode and cathode. Some types of fuel cells also contain corrosive chemicals in the electrolyte and so they must be handled and disposed of safely at the end of their working life.
Hydrogen is highly flammable 🔥 so storing and transporting it safely is another challenge. It can be stored as a compressed gas or as a liquid but both methods will require specialised tanks and infrastructure which will also be expensive. Converting existing petrol stations to supply hydrogen would also be extremely expensive and would require large-scale storage and pipe based transport systems would also need to be developed.
That said fuel cells have some clear advantages. They have no moving parts, which makes them reliable and efficient. They can provide a reliable source of electricity for remote communities and businesses.
When hydrogen is produced using low-carbon electricity fuel cells can be a much more environmentally friendly option than traditional methods used to generate electricity. A hydrogen-oxygen fuel cell produces water as the only waste product at the point of use. It does not release carbon dioxide, sulfur dioxide or nitrogen oxides while operating unlike burning fossil fuels.
So overall fuel cells are not a perfect solution yet; but if the hydrogen gas used in the fuel cells can be produced in a sustainable way then fuel cells could play an important role in reducing emissions in the future.
Below is a 6 mark exam style question on fuel cells and fossil fuels. Try the question yourself then compare your answer to the model one provided.
Hydrogen fuel cells are often suggested as a replacement for petrol and diesel engines. Evaluate the advantages and disadvantages of hydrogen fuel cells. Your answer should include environmental, economic and practical considerations. (6 marks)
Question (5–6 marks) – balanced, well-explained answer:
A high-level answer should clearly explain both advantages and disadvantages and reach a balanced conclusion.
⭐ Example concluding sentence:
"Hydrogen fuel cells have significant environmental advantages, but their overall sustainability depends on how the hydrogen is produced and whether costs can be reduced."
Another common exam question which is often set is to compare and evaluate the use of fuel cells and rechargeable batteries. The table below gives the main points which will help you answer these types of questions. Why not summarise these points in a set of flashcards listing the advantages and disadvantages of fuel cells and rechargeable batteries.
| Feature | Hydrogen Fuel Cells | Rechargeable Batteries |
|---|---|---|
| 🔋 How energy is supplied | Require a continuous supply of hydrogen and oxygen. Will produce electricity as long as fuel is supplied. | Use the store chemical energy inside the battery. Must be recharged once discharged. |
| ⛽ Refuelling / Recharging | Can be refuelled quickly, similar to filling a petrol car. | Charging can take much longer, depending on the battery size. |
| 🌍 Environmental impact (at use) | Produce only water at the point of use. No carbon dioxide is produced during operation. | Do not produce emissions during when in use. |
| ⚡ Overall environmental impact | Depends on how the hydrogen is produced. If it is made using fossil fuels then emissions may still be high. | Mining materials (e.g. lithium) can have serious environmental impacts. Electricity used for recharging may come from the burning of fossil fuels which means overall the harmful emissions will be high . |
| 💰 Cost | Currently expensive due to costly catalysts and new infrastructure. | Generally cheaper, though batteries for electric cars are still expensive and already widely available. |
| 🔧 Infrastructure | Requires new hydrogen production, storage and refuelling stations. | Charging points already widely installed and expanding. |
| 📏 Range | Often longer driving range before refuelling. | Range depends on battery size. Large batteries increase weight and cost. |