

Higher and foundation tiers
Chemically the alkanes are pretty dull and uninteresting. They are generally very unreactive molecules; the main use for them is as fuels. They are used to fuel the world! In fact alkane molecules provide the fuel for cars, trains, ships, planes and pretty much any other form of transport you can think of; other than electric vehicles. In this page we will discuss the products produced by the combustion of hydrocarbons such as the alkanes. Before we look at the combustion reactions of the alkanes consider the following two combustion reactions below:
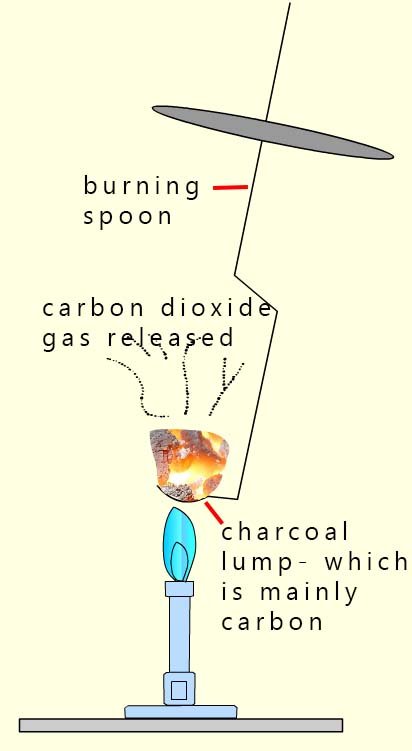
1. Carbon burns in air to produce carbon dioxide gas. The image opposite shows a lump of charcoal; which is mainly carbon burning in air to form carbon dioxide gas.
Equations for this reaction are shown below:
2. Hydrogen gas burns in air to produce water (water vapour).
Since alkanes are hydrocarbons and hydrocarbon molecules contain only the elements carbon and hydrogen, so when a hydrocarbon is combusted (burned) in air the products should be carbon dioxide and water. Essentially when you burn or combust a substance you take each element present in the compound and join it with oxygen.

3. Methane (CH4) is the gas used in Bunsen burners and at home for cooking and heating. It is also a hydrocarbon; so when it combusts in air it produces carbon dioxide and water vapour:
The gas we use at home for cooking and heating is methane. Methane is also used in the science lab as the fuel for Bunsen burners. If burning the hydrocarbon methane forms carbon dioxide and water vapour; then burning any hydrocarbon will also produce carbon dioxide and water vapour; for example the hydrocarbon pentane (C5H12) burns or combusts according to the equation below:
The only difference between burning pentane and methane is that pentane is a larger molecule which contains more carbon and hydrogen atoms, so it requires more oxygen to burn or combust completely, and it produces more water vapour and more carbon dioxide gas.
Combusting fuels which require large volumes of oxygen to burn completely can sometimes be a problem, for example if you try to burn a fuel inside sealed containers such as engines or a boiler where there may be a lack of oxygen. Burning hydrocarbons where there is a plentiful supply of oxygen available results in the production of carbon dioxide gas and water vapour. This is called complete combustion.
However burning hydrocarbons where there is a limited supply of oxygen is called incomplete combustion and different products are obtained. These reactions also release less energy than complete combustion. The two equations below show the products formed by the complete and incomplete combustion of pentane.
For the complete combustion of pentane 8 moles of oxygen gas are required for every mole of pentane burned. However if pentane is burned in a limited supply of oxygen then the poisonous gas carbon monoxide (CO) can be produced instead of carbon dioxide, this is shown in the second equation above.
Methane gas is the fuel used in Bunsen burners in the science lab. The two common flames used on the Bunsen burner are the yellow safety flame and the blue roaring or heating flame. The safety flame is an orange/yellow visible flame produced when the air hole is closed, while the blue roaring flame is produced when the air hole is fully opened. Opening the air hole allows air to be drawn into the Bunsen chimney where it mixes with methane gas. This means that when the mixture of air and methane is lit at the top of the chimney there is enough oxygen present for complete combustion to take place.
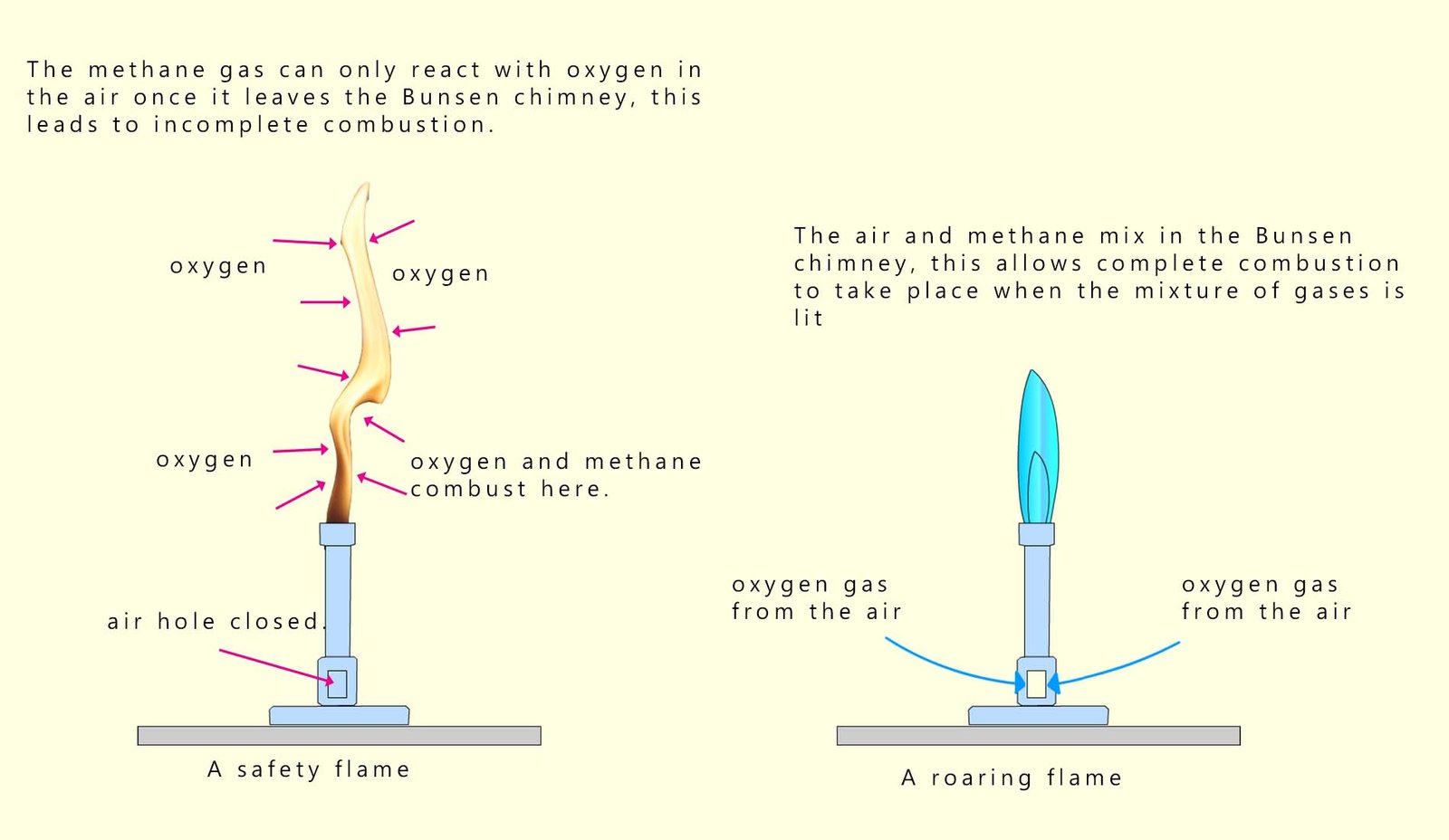
However if the air hole is closed then methane gas can only combust once it leaves the chimney and mixes with the air. This leads to incomplete combustion because there is not enough oxygen for the methane to react with. The luminous safety flame is due to soot or carbon particles in the flame; these particles reflect light and make the flame visible. This is outlined in the image shown.
The word and symbolic equations for these combustion reactions of methane are given below:
The incomplete combustion of methane produces carbon monoxide, whereas when methane is burned in a plentiful supply of oxygen it produces carbon dioxide.
So where does the soot come from in the luminous Bunsen flame? Well keep reading for the answer......
Click the correct statement(s) underneath each image of the Bunsen flame to correctly describe the safety and the roaring Bunsen heating flame.
Click the correct description for each flame.



Propane (C3H8) is another commonly used alkane. Propane is sold in gas bottles for use in BBQs, heating homes and caravans; it is also commonly used in the construction industry. The equations below show the products of the complete and incomplete combustion of propane.
It is a similar story when propane is burned: the complete combustion of a hydrocarbon fuel like propane produces carbon dioxide and water vapour, whereas incomplete combustion produces the toxic gas carbon monoxide and water vapour.
Note: in some of the equations above (for example, the incomplete combustion of methane and propane) I have used 11/2 and 31/2 moles of oxygen to balance the equations. In some textbooks you may see the equations doubled to remove the fractions. The choice is yours!
If hydrocarbons are burned in a very limited supply of air/oxygen it is possible that carbon monoxide and solid carbon (soot) will be produced. The equations below show the complete and incomplete combustion of propane (C3H8) when various amounts of oxygen are available. You can see that when the oxygen supply is very limited then carbon (soot) is formed.
(Hint: to balance these equations for the incomplete combustion, however many carbon atoms are in the hydrocarbon that will be the number of moles of soot produced and however many hydrogen atoms are present in the hydrocarbon, half that number of moles of water will be produced.
It is very likely that when hydrocarbon fuels undergo combustion a combination of carbon dioxide, carbon monoxide and soot can all be produced. It all depends on the amount of oxygen available when the fuel is burned. The equations below show the complete and incomplete combustion of hexane (C6H14):
Click the oxygen slider to move it from very limited oxygen to limited oxygen and then to plentiful oxygen and in each case try to predict the products that are formed when a hydrocarbon fuel is burned.
Change the amount of oxygen available. Watch how the products of combustion change.
As discussed above the products of complete combustion of a hydrocarbon are carbon dioxide and water. The apparatus below can be set up to collect the products of combustion of a hydrocarbon and test them.
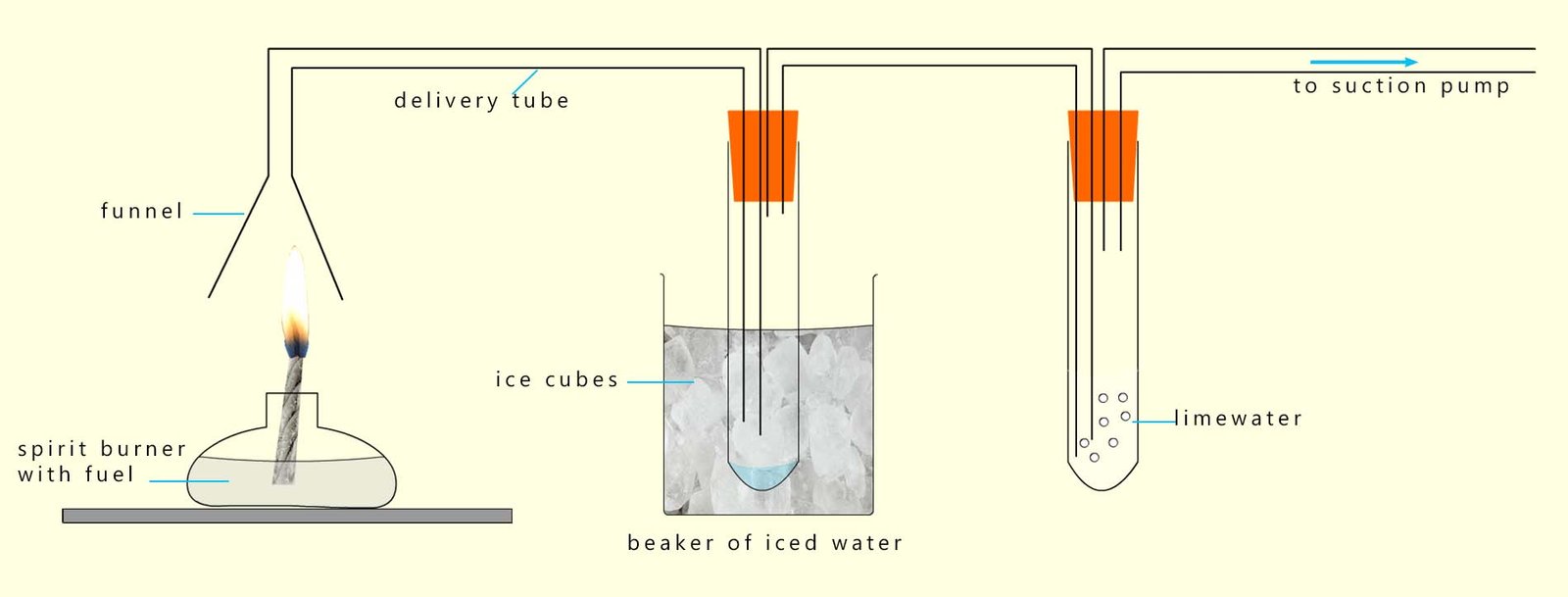
The products of complete combustion of a hydrocarbon fuel are
carbon dioxide and water vapour.
These two gases leave the candle flame and are drawn into the funnel by a suction pump.
The vapours then enter the first boiling tube; which is surrounded by ice-chilled water.
This causes the water vapour to condense and collect as liquid water.
This can then be tested with blue cobalt chloride paper
(blue cobalt chloride paper turns pink in the presence of water).
Alternatively the boiling tube can be filled with anhydrous copper sulfate, a white solid which turns blue when
water is added.
Match the terms below with their correct definition by clicking the term and then its correct definition. Correct responses will turn the definitions green!
As mentioned above the main use for alkanes is as fuels for transport, heating and industrial uses. Many fuels not only contain alkanes but also the element sulfur; which is present as an impurity. Sulfur is a yellow solid in its elemental form. However it forms the acidic non-metal oxide sulfur dioxide when it is burned:
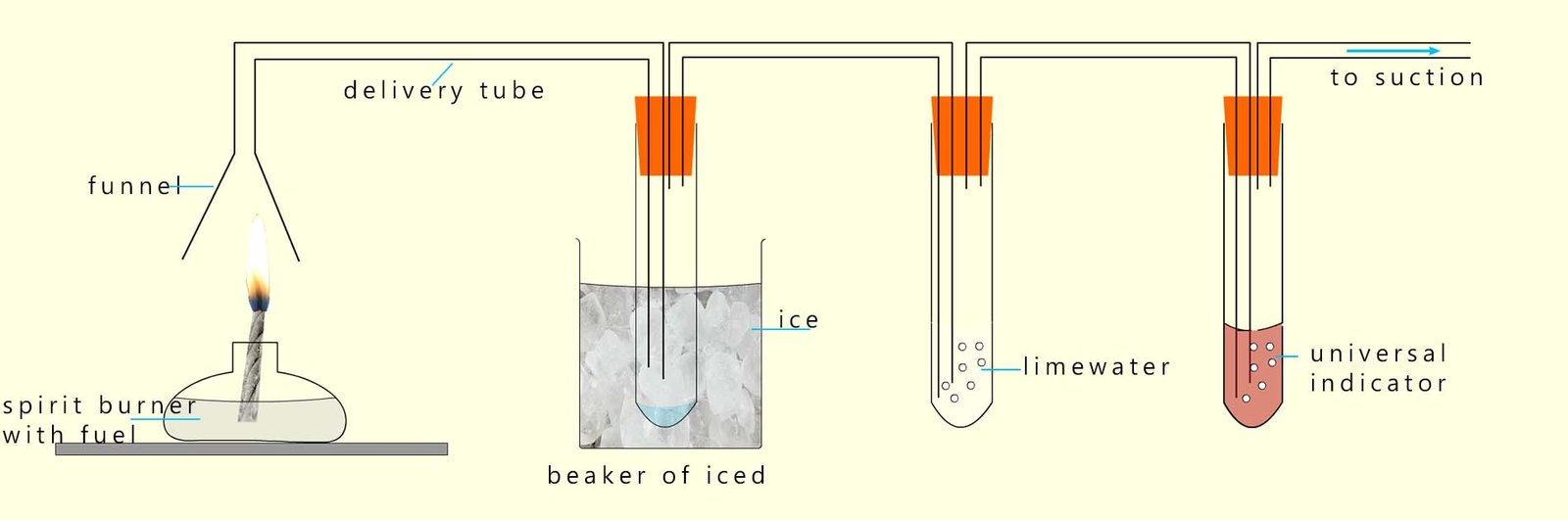
Sulfur dioxide is an acidic gas and dissolves in water to form an acid (sulfurous acid). We can modify the apparatus above to test for this acidic sulfur dioxide gas by adding another boiling tube filled with universal indicator solution. If acidic gases are present; such as sulfur dioxide then the universal indicator solution will turn red. This new set-up is shown below:
Review your understanding of the main terms and ideas on combustion reactions by completing the two paragraphs below using the words from the drop-down menus. To help you select the correct words, all the words in the drop-down menus are shown in the yellow boxes below.
Why not use the summary table below to produce a set of revision flashcards or revision notes before attempting the exam style questions below?
| Topic 🧠 | Key idea ✅ | Example / test 🧪 |
|---|---|---|
| Complete combustion 🔥 | Burning a hydrocarbon in a plentiful supply of oxygen produces carbon dioxide and water vapour. |
Example: methane CH4 + 2O2 → CO2 + 2H2O |
| Incomplete combustion ⚠️ | Limited oxygen means the fuel can’t fully burn, so you can get carbon monoxide and/or soot (carbon) as well as water vapour. |
Example: methane CH4 + 11/2O2 → CO + 2H2O |
| Why it matters 🚗🏠 | Incomplete combustion wastes fuel (less energy released) and produces toxic gases such as carbon monoxide. | Often happens inside engines/boilers where oxygen supply is limited. |
| Test for water 💧 | Water forms during combustion and can be confirmed with a simple chemical test. | Blue cobalt chloride paper turns pink in the presence of water. |
| Test for carbon dioxide 🫧 | Carbon dioxide is a product of complete combustion. | Limewater turns milky/chalky when CO2 is bubbled through it. |
| Sulfur impurity & acid rain 🌧️ | Some fuels contain sulfur as an impurity; burning it produces an acidic gas. |
S + O2 → SO2 SO2 dissolves in water and contributes to acid rain. |
Identify what is wrong in each of the statements below:
Each statement below contains one common mistake. Click the option that explains what is wrong.