
Higher and foundation tiers
Organic chemistry is probably one of the largest and perhaps most exciting areas of study in chemistry. It is concerned with the study of compounds which contain the element carbon. Hydrocarbons are organic compounds made up of molecules containing only the elements hydrogen and carbon. Hydrocarbons are found in crude oil.
Crude oil is a natural substance; it is a thick, sticky substance which consists of a mixture of many thousands of different hydrocarbon molecules. Crude oil is often described as the "lifeblood of industrial nations" because of the many valuable substances it contains. Crude oil is a very valuable natural resource which is extracted from under the oceans and on land in many countries spread over the globe including: Venezuela, Saudi Arabia, Iran, Iraq, UAE, Russia, China, Brazil, Canada as well as many others.
Hydrocarbons are everywhere around you; they are in fuels, plastics, gases for heating and cooking and even in many everyday products. Most of these useful substances originally come from crude oil. On this page you’ll learn what hydrocarbons are, how crude oil was formed and why the simplest family of hydrocarbons; the alkanes are so important in GCSE Chemistry.
By the end of this page, you should feel confident explaining what saturated means, recognising members of the alkane homologous series and using the general formula CnH2n+2.
Crude oil is a fossil fuel formed from the remains of microscopic sea organisms called plankton. When these plankton died they were buried in sediments on the ocean floor. Being buried meant there was little or no oxygen, so decay by bacteria was greatly reduced. Over millions of years layers of sediment built up, causing an increase in temperature and pressure which effectively "slow-cooked" the remains and turned them into crude oil and natural gas. Since crude oil takes millions of years to form and we are using it up faster than it can be replaced, it is a finite resource and will eventually run out.
Try the activity below to test your understanding of how crude oil forms.
Click a card, then click the bin you want to drop it into (no dragging). If you change your mind, just clear a bin and try again.
Most of the compounds found in crude oil are hydrocarbons. There are thousands of different hydrocarbon molecules and to make it easier to name and identify them they are placed in groups or families where members of each family have something in common. The simplest family of hydrocarbons is called the alkanes. The alkanes are hydrocarbons which have only single covalent bonds present. The first four members of the alkane family or homologous series are methane, ethane, propane and butane. They are shown in the image below.
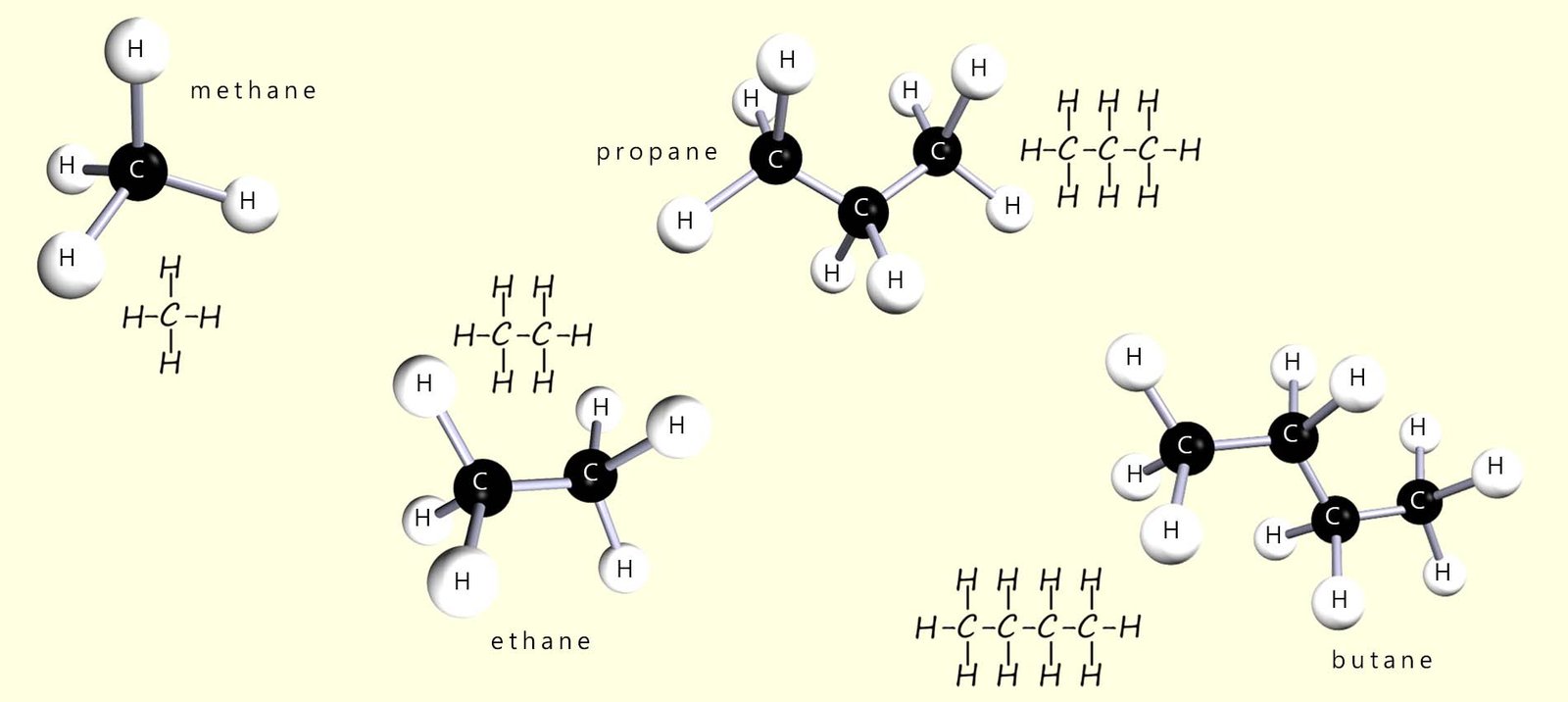
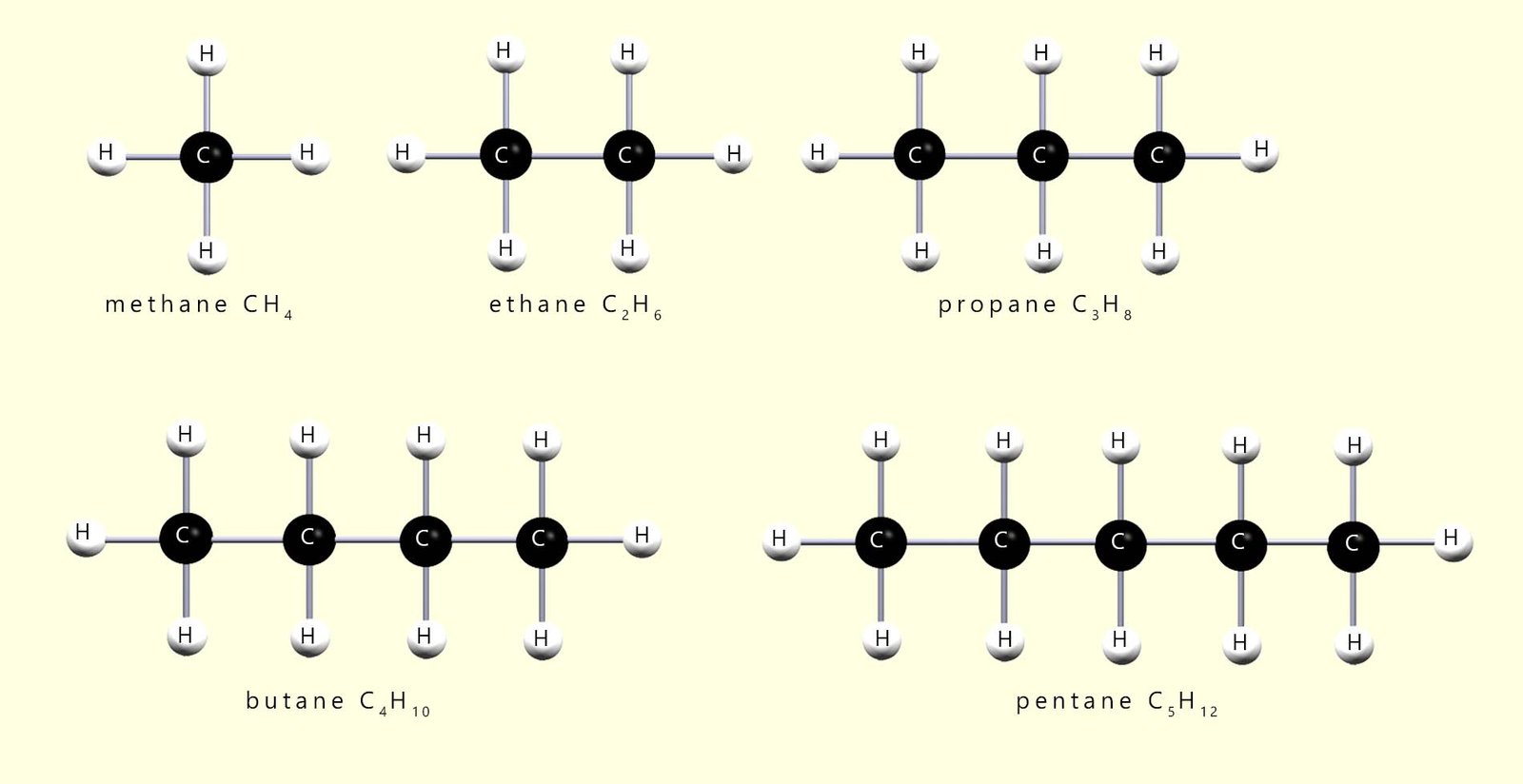
The alkanes shown in the image below have a backbone of carbon atoms surrounded by a “sea” of hydrogen atoms. Each carbon atom forms four covalent bonds and each hydrogen atom forms one covalent bond. The alkanes are described as saturated because there are only single covalent bonds between the carbon atoms. The structures above are shown as 3D models to represent their real shape, but they are often drawn as flat 2D structures. At GCSE level you are not usually expected to worry about the exact 3D shape and most exam questions use flat displayed formulae. The first five alkanes are shown below in this 2D form, although in reality they are three-dimensional.
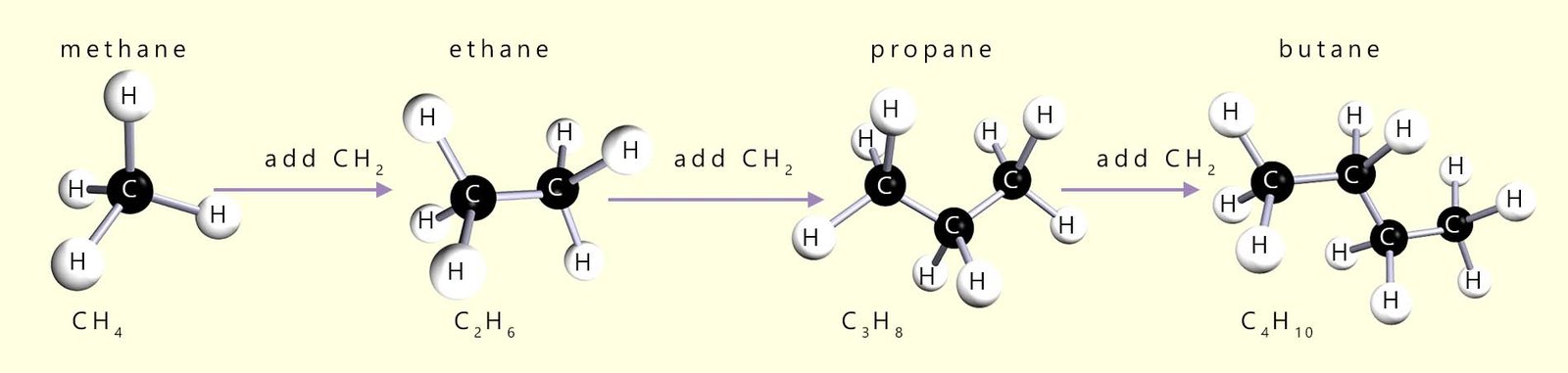
If you study the formula for each of the alkane molecules above you will notice that in going from one alkane to the next we simply add a -CH2- group each time:
The alkanes form a homologous series; that is, a series of compounds which all have the same general formula and which show trends in their physical properties, while having similar chemical properties. You can work out the formula for any alkane molecule using the general formula CnH2n+2. As an example, consider decane which is an alkane with 10 carbon atoms. Substitute 10 into the general formula: (2 × 10) + 2 = 22, so the molecular formula for decane is C10H22.
Match the terms below with their correct definition by clicking on the term and then its correct definition. Correct responses will turn the definitions green!
The names for all members of the alkane homologous series (family) end in -ane. The first part of the name (the prefix) tells you the number of carbon atoms present. The table below shows the first ten alkanes and their molecular formulae.
| alkane | molecular formula |
|---|---|
| 1️⃣ methane | CH4 |
| 2️⃣ ethane | C2H6 |
| 3️⃣ propane | C3H8 |
| 4️⃣ butane | C4H10 |
| 5️⃣ pentane | C5H12 |
| 6️⃣ hexane | C6H14 |
| 7️⃣ heptane | C7H16 |
| 8️⃣ octane | C8H18 |
| 9️⃣ nonane | C9H20 |
| 🔟 decane | C10H22 |
Once you become more familiar with organic compounds you will learn that the name often tells you the number of carbon atoms present:
From the table above you should be able to see that hex- means 6 carbon atoms, hept- means 7 carbon atoms and oct- means 8 carbon atoms. The ending -ane shows the molecule is an alkane, which means it is a saturated hydrocarbon.
Complete the paragraphs below by filling in the blanks to summarise the main points above. The words to complete the sentences are shown in the yellow boxes; select the correct word from the drop-down menus.